 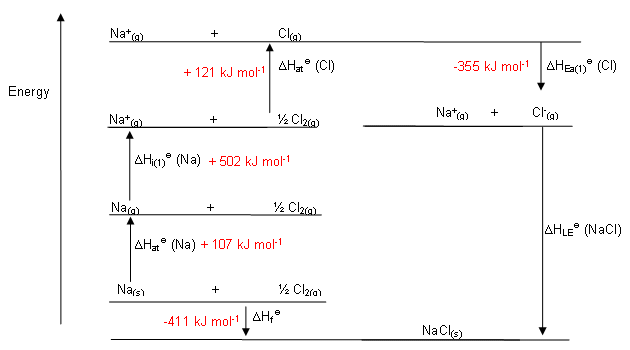
Ionization Energy is the amount of energy required to remove an electron from a neutral atom or ion. This process always needs an energy input and hence has a positive value. Let us understand these concepts in more detail. Hess' Law is effectively applied to an ionic solid in the Born Haber Cycle.īefore the Born-Haber Cycle can be used to calculate the lattice energy of ionic solid, some concepts mentioned below need to be understood: This lattice energy can only be approximately measured through Hess’s law.Īccording to Hess's Law, the overall change in energy of a process may be calculated by breaking the process down into parts and then summing the energy changes of each phase. It is defined as the energy released when gaseous ions are from a mole of a solid ionic compound or as the energy that is required to convert one mole of ionic solid into its gaseous ions. Lattice energy keeps together the anions and cations of the compound in fixed positions in a crystalline solid. This difference is due to the difference in enthalpy known as ‘Lattice energy’, between the ionic solids. Due to the stability of the electrostatic force of attraction between positive and negative charges, ionic compounds are expected to have identical physical properties. However, their physical properties such as water solubility and stability differ. Alkali and alkaline earth metals react with the elements of the halogen family or chalcogen to form compounds that are crystalline ionic solids. Ionic solids are produced by the reaction of electropositive metals along with electronegative nonmetals. Its values are often given in kJ/mol units. Through this definition, the process will always be exothermic, and therefore the value for lattice energy will be negative. The other definition of lattice energy defines is as the energy produced when gaseous ions bond to create an ionic solid. As this is an endothermic process, the value for the lattice energy is always positive. Lattice Energy is a form of potential energy that is necessary to break apart an ionic solid and transform its component atoms into gaseous ions. 9.The enthalpy change involved in the production of ionic solids from gaseous ions, or the energy involved in breaking the ionic solids into gaseous ions, is referred to as the lattice energy. A Born-Haber cycle calculates the lattice energy using Hess' law by comparing the standard enthalpy change in the production of an ionic solid to the enthalpy required to produce gaseous ions from the elements. Born Haber cycle is used to determine the lattice energy, electron affinity, and crystal energy.Calculate the lattice energy for CaF 2.Įnergy of formation for one mole of CaF 2 from its elements = -1228 kJ/molīack to Periodic Trends and Ionic Compounds The sublimation energy for Sr is 164 kJ/mol, E i1 = +549.5 kJ/mol, E i2 = +1064.2 kJ/mol, E ea for Cl (g) = -348.6 kJ/mol, energy for the formation of one mole of SrCl 2 from its elements = -803.7 kJ/mol, and the bond dissociation energy for Cl 2 (g) = +243 kJ/mol.Įxercise 5. Which has the highest lattice energy, Na 3P or Na 2S?Įxercise 4. Order the following ionic compounds from lowest to highest lattice energy. What is the lattice energy for MgF 2?Įxercise 2. Consider the following Born-Haber cycle for MgF 2. Worksheet: Ionic Compounds, Born-Haber Cycle, and Lattice EnergyĮxercise 1. A more extensive list can be found in other tables or in the Handbook of Physics and Chemistry. 
In the case of NaCl and MgCl 2, the MgCl 2 has the larger lattice energy because the magnesium cation is smaller, and the charge is a +2 rather than a +1 for the sodium ion.īelow is a table with the lattice energies of some ionic compounds. This is because the charges are the same, but the potassium ion is larger than the sodium ion. For example, the lattice energy of NaCl is larger than the lattice energy of KCl. Lattice energies are large when ions are closer together, the distance between the ions is small and when the charges are larger. Coulomb’s law is equal to a constant, k, multiplied by the product of the ion charges, z 1 and z 22, between the ions. Recall, lattice energy is positive meaning it is endothermic. The stronger the bond, the higher the lattice energy. Lattice energy, E lattice is dependent on the strength of the bond between the cation and anion in an ionic bond. The lattice energy is always positive, because it takes energy to separate the ions from the solid. 
The equation for the lattice energy is the reverse of the equation in Step 5 in the figure below, for the formation of the solid from its ions which releases 787 kJ/mol of energy.Ī Born-Haber cycle allows the calculation of the lattice energy for a solid ionic compound. The process absorbs energy, and is highly endothermic. Lattice energy, E lattice is the energy required to separate one mole of a solid ionic compound into its gaseous ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
